Background:
Background:
Cosmetic firms are responsible for substantiating the safety of their products and ingredients before marketing. Failure to adequately substantiate the safety of a cosmetic product or its ingredients prior to marketing causes the product to be misbranded unless the following warning statement appears conspicuously on the principal display panel of the product's label:
"Warning--The safety of this product has not been determined." (21 CFR 740.10)
In addition, regulations prohibit or restrict the use of several ingredients5 in cosmetic products and require warning statements6 on the labels of certain types of cosmetics.
In general, except for color additives and those ingredients which are prohibited or restricted from use in cosmetics by regulation, a manufacturer may use any ingredient in the formulation of a cosmetic provided that the ingredient and the finished cosmetic are safe, the product is properly labeled, and the use of the ingredient does not otherwise cause the cosmetic to be adulterated or misbranded under the laws that FDA enforces.
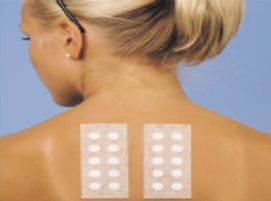
Sensitization is the process by which humans can develop over time increased allergic responses to a substance through repeated exposure to that substance. It is different from irritation because it involves the immune response. Due to immune response, the reaction becomes worse with repeated exposure, and it is usually specific to individuals. The HRIPT consists of 2 phases, and sometimes 3.
Phase I is the Induction Phase where the product is applied to the skin 9 times during the course of 3 weeks. This is followed by a two-week rest period after which the skin is exposed to the product again in Phase II or the Elicitation Phase. A response in Phase II is usually allergic in nature and Phase III is used to verify and better define the reaction. There are different methods that are available which have different application phases but the resulting predictions of allergy and irritation response are the scientific goals. The industry standard for these tests is usually to use a minimum of 50 human volunteers. However, up to 200 subjects is sometimes used for better statistical prediction. Dermatologist involvement in these studies as signatory or Primary Investigator is also offered. Safe for Sensitive Skin is a common claim whereby subjects are recruited that either have self perceived sensitive skin or test positive using the lactic acid sting test.
Other tests available:
Phase I (Cutaneous Safety Tests)
- 14 day and 21 day cumulative skin irritation test
- 24 hour and 48 hour patch tests for acute skin irritation (primary irritation)
- Human Repeat Insult Patch Test (HRIPT)
- Modified maximization test
- Photoallergy Maximization Test
- Phototoxicty Test
Photosensitization
Photosensitization can occur when normally harmless molecules undergo changes when exposed to UV light. The HRIPT method is modified in combination with exposure to UV-A irradiation to determine the phototoxic or photoallergic potential of a topically applied product.